The Parsemus Foundation works to create meaningful improvements in human and animal health and welfare by advancing innovative and neglected medical research. The foundation supports proof-of-concept studies and then seeks to raise awareness of results, to ensure that they change treatment practice rather than disappear into the scientific literature. Many of the studies the foundation supports involve low-cost approaches that are not under patent, and are thus unlikely to be pursued by pharmaceutical companies due to limited profit potential.
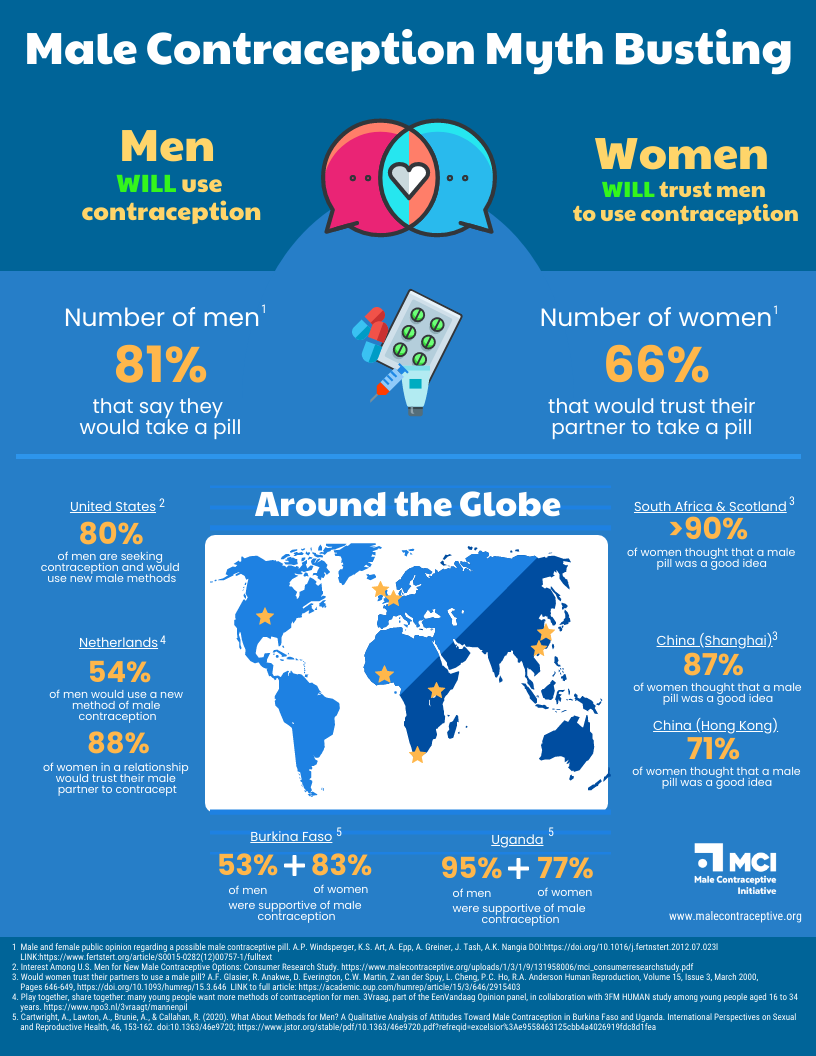
The Parsemus Foundation’s main focus has been developing Vasalgel as a non-hormonal male contraceptive. The foundation has partnered with NEXT Life Sciences to complete development of the product and bring it to market as Plan A. For information on Vasalgel, please contact NEXT Life Sciences.
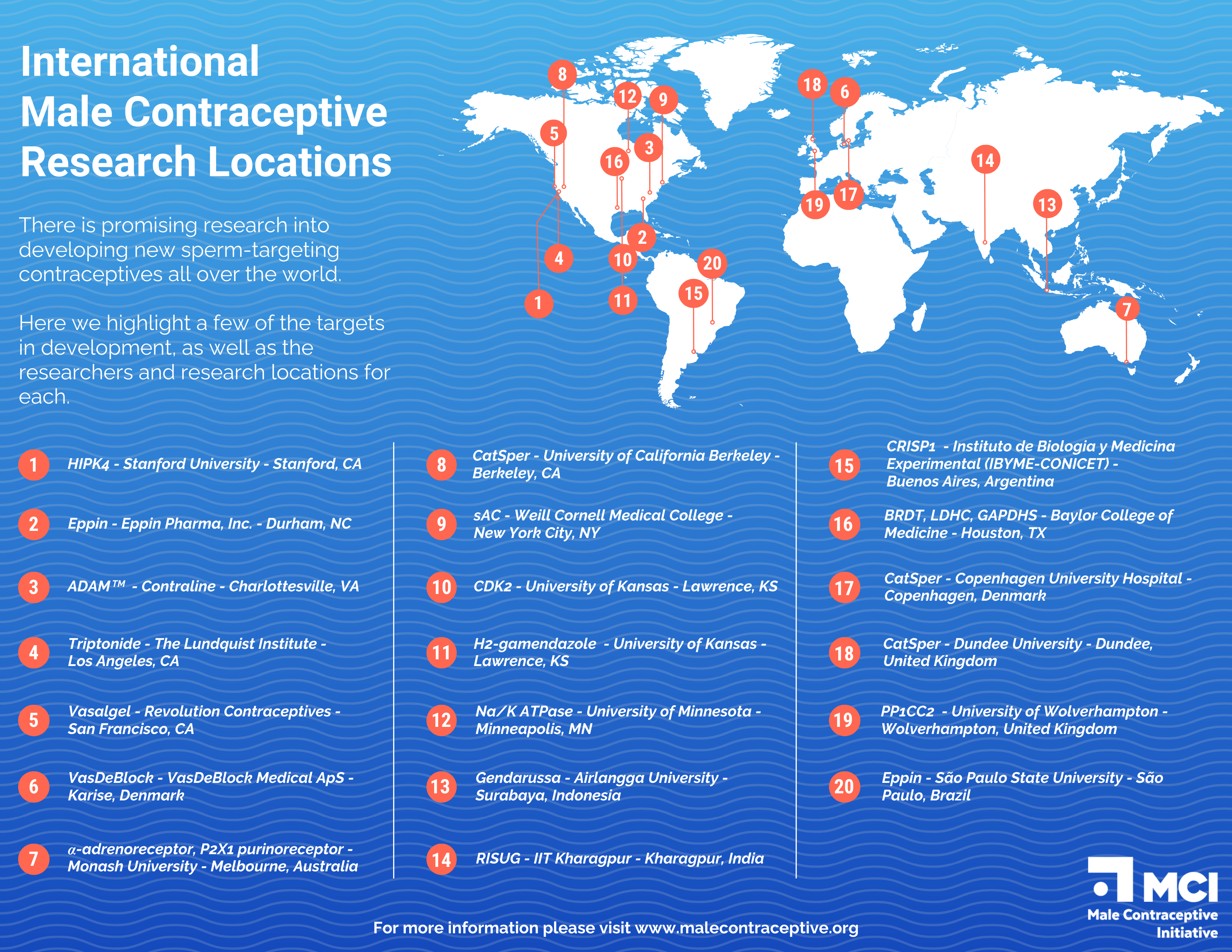
For information on the development of other non-hormonal male contraceptives, visit the Male Contraceptive Initiative.
The foundation also works on pet contraception, including hormone-sparing sterilization options and hormone restoration; noninvasive treatment for benign prostatic hyperplasia (enlarged prostate); prevention of dementia and Alzheimer’s disease; and iron deficiency.
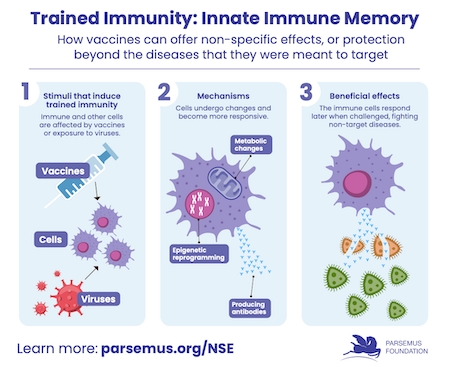
The Parsemus Foundation supported clinical studies on the effectiveness of existing medications to treat COVID infection, including enrollment support for the University of Minnesota’s COVID-Out trial.
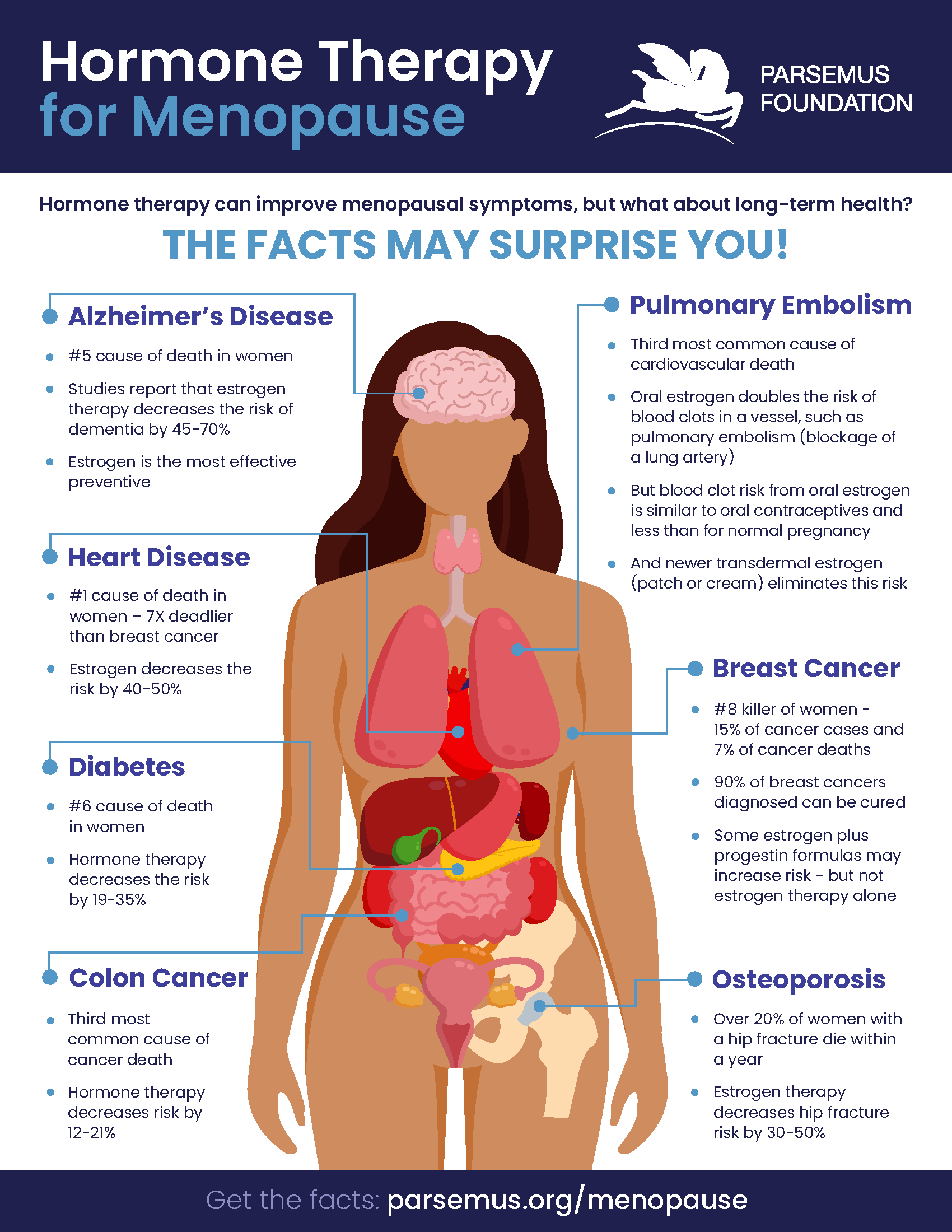
The foundation has also supported the development of the nonprofit Hormonally to ensure that evidence-based information about menopause hormone therapy is available to women and physicians.
For media coverage of our projects, please contact us at media@parsemus.org